Introduction
Although progression free survival and overall survival of patients with Hodgkin lymphoma (HL) has improved in the past 10 years, 15 % of patients will fail to conventional therapy and die of their lymphoma. The search of new prognostic factors for identifying these high risk patients at diagnosis of HL remains challenging in PET-guided strategies developed to improve the outcome HL patients while reducing treatment toxicity
The aim of this study was therefore to assess the impact of baseline tumor microenvironnement in a large cohort of HL patients prospectively treated with upfront escalated BEACOPP in a randomized phase III clinical trial evaluating a PET-driven strategy (AHL 2011, NCT01358747) and to compare its prognostic value with other clinicopathological markers.
Material and Methods
Tumoral material was collected from HL patients prospectively enrolled in the AHL2011 study from LYSA centers in France and Belgium. The AHL2011 study was designed to evaluate in 16-60y HL patients with Ann Arbor stage III, IV or high risk IIB, a de-escalade PET-driven strategy after 2 cycles of BEACOPPesc (PET2) randomly compared to a standard treatment not driven by PET delivering 6 cycles of BEACOPPesc. The AHL2011 strategy was shown to safely guide treatment of advanced HL patients allowing using ABVD in early responders to BEACOPPesc (PET2 negative) reducing toxicities without impairing disease control (Casasnovas et al, Lancet Oncol 2019, JCO 2022).
The tumor microenvironnement (TME) was analyzed on formalin fixed paraffin embedded diagnostic tissue biopsy obtained before treatment by morphological evaluation on standard staining on whole slides (% polynuclear eosinophils, % lymphocytes, % plasmocytes, % histiocytes), and immunohistochemistry (IHC) scoring (score 0-1-2-3) for CD20, CD3, CD4, CD8, CD68-TAMs (tumor-associated macrophages), CD163 (n=680) and EBERS (n= 634). Additional staining were done on Tissue microarray (TMA n=409) for PD-1, PD-L1, TIM3, GATA3, CD45R0, TBET and were centrally reviewed. Computational analysis was used for the TMA analysis using Tissu Studio software (Definiens). For each marker, a H-score was calculated (stained cells x 1, 2 or 3 according to the intensity of staining divided by the total number of cells x100)
Results
680 patients were included on the whole slides analyses and 409 for those on the TMA analyses. Patients included had similar characteristics to the remaining patients of AHL2011 not included here. Among all the microenvironnement biomarkers tested only CD20 and CD68 had prognosis value.
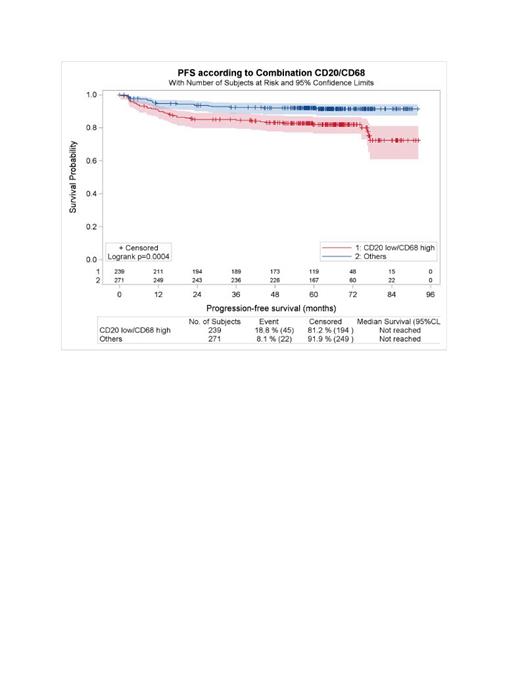
In univariate analysis, high CD20 H-score was associated with a favorable PFS (HR=0.43, 95%CI 0.22-0.86), while patients having high CD68 H-score had unfavorable PFS (HR=2.29, 95%CI 1.29-4.05). Then, the patients with a combined CD20 low/CD68high TME (n= 239, 47%) had a significant lower PFS compared to other groups (HR=2.46, 95%CI 1.48-4.09; p=0.004) (figure 1).
In multivariate analysis including all relevant variables (Bulky disease / IPS / TMTV (total metabolic volume) / PET2 /PET4 / combination CD20 CD68 / % of tumor cells /EBV status / B symptoms), only High TMTV >220ml (HR = 2.09, p =0.017), PET4 positivity (HR= 7.06 ; p < 0.0001) and the CD20 low/CD68high profile (HR=1.95, p=0.025) had an independant prognosis value on PFS.
Patients harboring both a TME depleted in CD20 and enriched in CD68, had peculiarities. Indeed, they had significantly more B symptoms (78.2% vs 64.2%, p<0.001), higher median TMTV (270 vs 163ml, p<0.001), were more frequently EBERs positive (23.6 vs 12.7%, p=0.002), had more frequently lymphopenia (38.5 vs 18.2%, p<0.001) and high IPS (69.2 vs 53%, p<0.001). Finally, patients with a CD20low/CD68high profile, had a higher median tumor cells PD-L1 H-score compared to other patients (39.7 vs 7.4, p<0.001).
Conclusion
Altogether these results demonstrate in a large PET-guided phase III trial (AHL2011), the interest of analyzing CD20 and CD68 expressions within the tumor microenvironnement to better evaluate the risk of treatment failure prior to therapy in patients with advanced HL. The prognosis value of the CD20/CD68 profile is independant of those of the baseline PET metrics (TMTV), and the early reponse to treatment. The enrichment of highly positive PD-L1 tumor cells in patients with CD20 low/CD68high TME suggests that PD-1 blockers could be a good therapeutic option in frontline treatment of this poor prognosis group.
Disclosures
Ghesquieres:Gilead, Roche: Consultancy; Gilead, Roche, Bristol Myers Squibb, AbbVie, Novartis: Honoraria. Casasnovas:BMS: Consultancy, Honoraria; MSD: Consultancy, Honoraria; ADC therapeutics: Consultancy, Honoraria; Takeda: Consultancy, Honoraria, Research Funding; Roche: Consultancy, Honoraria, Research Funding; Beigene: Consultancy, Honoraria; Kite/Gilead: Consultancy, Honoraria, Research Funding; Astra Zeneca: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal